MAIN TOPIC: RADIOPHARMACEUTICALS ON THEIR WAY TO THE PATIENTS
A seven-year development plan of Russian pharmaceutical industry (Pharma 2030) includes all the tools that would allow Russia to implement technological self-sufficiency in pharmaceutical industry, in particular in development, research, and production of radiopharmaceuticals. Dmitry A. Rozhdestvensky, Cand. Sci. (Med.), Coordination manager for circulation of medicinal products and medical devices, Technical Regulation and Accreditation Department of the Eurasian Economic Commission, shares his insights on how to develop, produce, authorise, and apply radionuclide therapy in the Member States of the Eurasian Economic Union (EAEU).
INTRODUCTION. Radiopharmaceuticals play a crucial role in modern nuclear medicine, offering effective methods for cancer diagnostics, treatment, and theranostics. Despite the rapid growth, the field still lacks a comprehensive patent analysis that would identify key scientific and technological achievements, market leaders, and potential risks and opportunities. Systematising and analysing patent activity for radiopharmaceuticals will help both track the evolution of technology and identify promising areas for future research and commercialisation.
AIM. This study aimed to analyse patent activity comprehensively in order to assess its current state and prospects of radiopharmaceutical industry.
MATERIALS AND METHODS. The study was performed using Questel Orbit database. Patent and bibliometric analysis, segmentation of scientific and technical information was used to analyse 4,988 patent families over a period of 2000–2024. Eligibility criteria included indexes of International Patent Classification and keywords in the Russian and English languages.
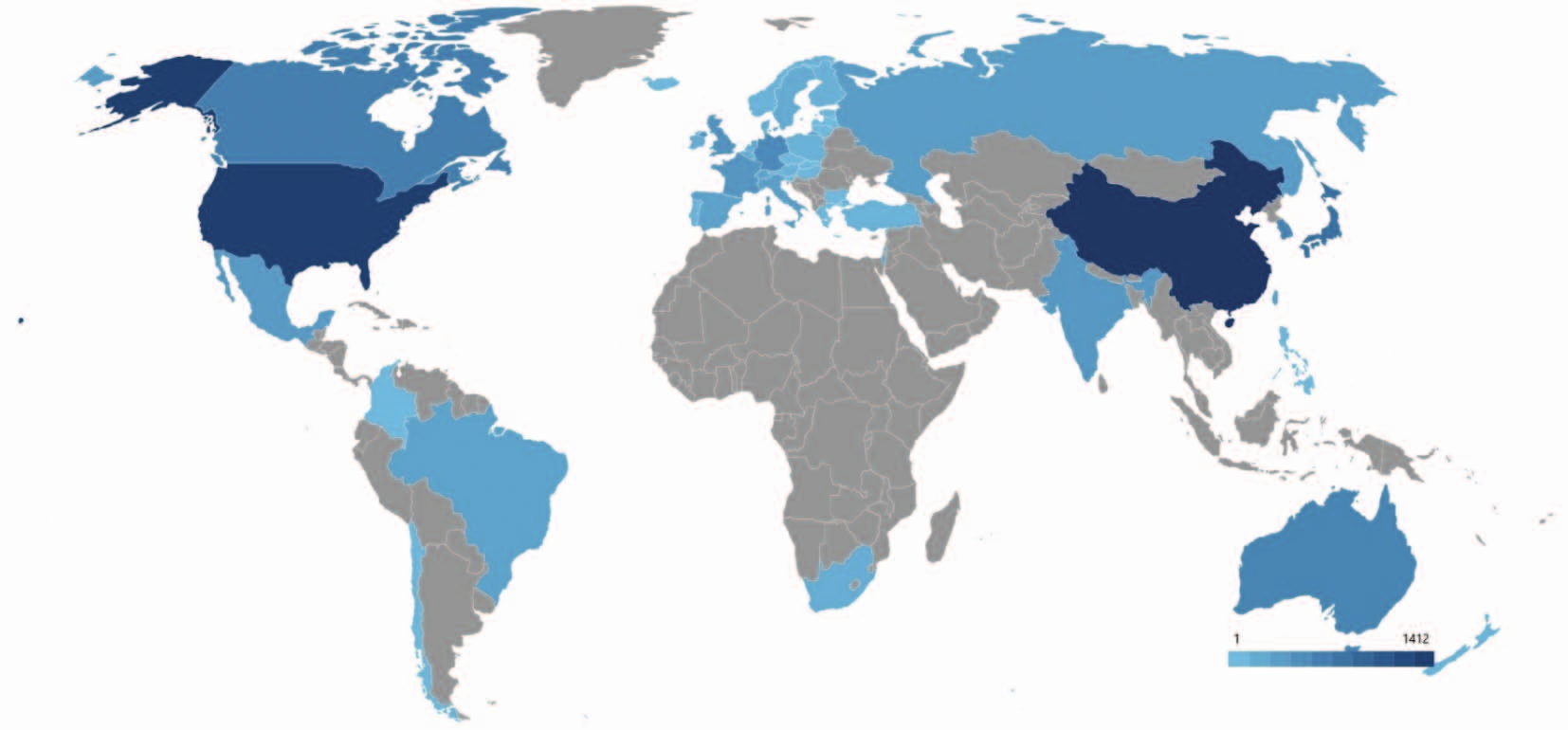
RESULTS. There has been an increase in patent activity after 2018 related to approval of innovative drugs based on 177Lu and 68Ga. Three main patenting areas were identified: diagnostics (43%), treatment (23%), and theranostics (34%). The leaders in the number of patents are the USA, China and the EU countries that have well-developed science and technology base and strong market potential. Differences were found in patent strategies of Russian and foreign organisations. Foreign institutions prefer using broad claims: one patent consolidates a whole class of radionuclides, several molecular carriers and delivery mechanisms, providing wide legal protection and protecting innovations in case of minor formula changes. This strategy allows for a complex protection of technological solutions and provides stable market advantage. At the same time, Russian companies prefer narrowly focused patents that cover specific process solutions. This allows protecting separate innovations, but makes broader coverage impossible and gives the competitors a chance to circumvent the patent.
CONCLUSIONS. Patent analysis shows the focus of developmental studies shifted from traditional diagnostic radiopharmaceuticals towards complex theranostic radiopharmaceuticals (with combined diagnostic and therapeutic properties). Despite available practical designs in radiopharmacy, Russia is far behind the leading countries in patenting. Moreover, the differences detected in patent claims of Russian and foreign scientists as well as patenting as a whole emphasise the need to improve patenting strategy in Russia in order to enhance intellectual property protection and stimulate international development of radiopharmaceuticals.
INTRODUCTION. The demand for new diagnostic and treatment solutions using radiopharmaceuticals (RPs), along with the growing nuclear medicine market, drives active development and rapid introduction of innovative RPs into circulation. These products possess a complex multi-component structure and specific radioactivity, a feature to be taken into account when planning and controlling research during drug development and subsequent technology transfer.
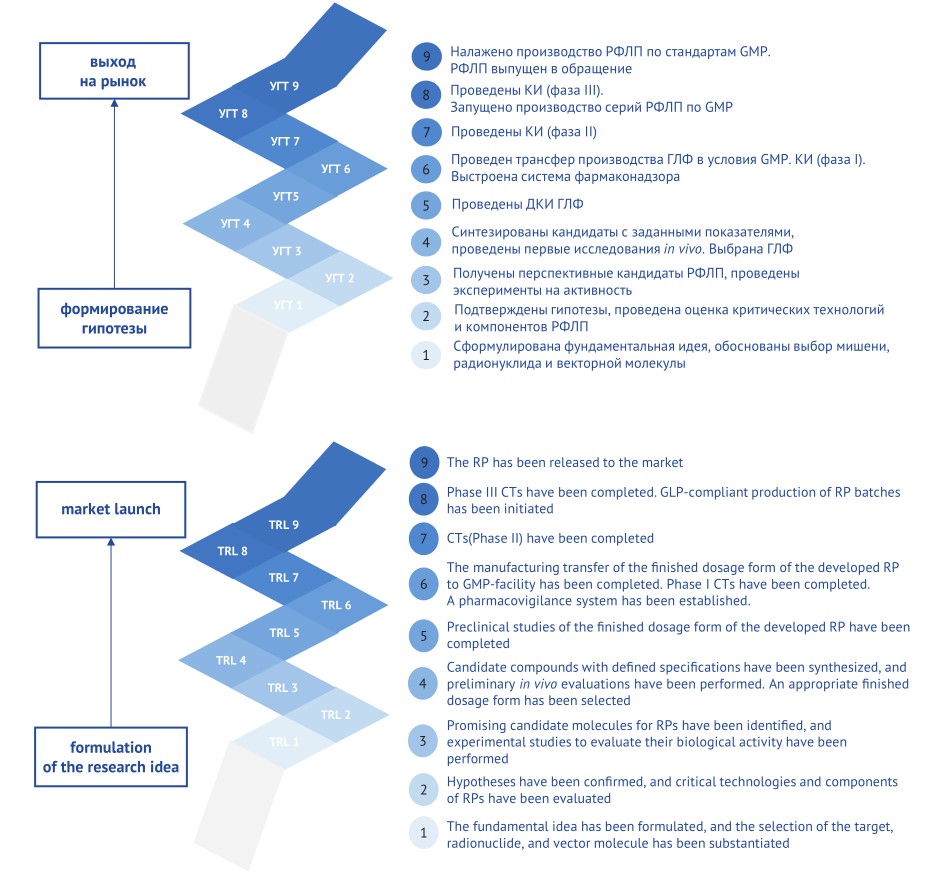
AIM. This study aimed to optimise RP development process using Technology Readiness Level (TRL) scale.
DISCUSSION. The authors analysed application of an industry-specific TRL scale for RPs. This scale considers key points for RPs: isotope content and biological molecules in the compound; necessity of quality control for specific parameters such as radiochemical purity, stability, and biological activity, as well as market circulation specifics. Correctly choosing intended use and marketing option of a developed RP is critically important, as this defines not only registration strategy but also quality requirements for the resulting product. Experience of JSC “Rosatom Science” has highlighted potential key risks, such as equipment contamination, radioactive decay rate during logistics, and other parameters. The key points in applying the scale to generic RPs were also high-lighted: the first four stages of the scale accelerated; necessity to select an original target molecule, as well as design differences of preclinical and clinical trials to obtain convincing evidence of generic bioequivalence to the reference product.
CONCLUSIONS. The adapted TRL scale for RPs is a validated tool that enables a comprehensive control over key development stages, timely risk identification and mitigation. The scale contributes to accelerating market introduction of innovative and generic RPs through transparent and stepwise planning, standard process monitoring, and use of the full development cycle, including marketing strategy.
INTRODUCTION. The concept of pharmacy preparation of radiopharmaceuticals (RPs) was first introduced in Russia with the entry into force of the Federal Law No. 61-FZ “On Circulation of Medicines” of 12 April 2010. Recently, significant legislative changes have been gained regarding RP production and preparation. Today it has become possible to make unregistered RPs at a medical organisation covering the in-house needs, which will allow to promptly advance new methods and means to treat malignant neoplasms as well as some other diseases.
AIM. This study aimed to analyse main growth vectors for Russian organisations planning to preparing RPs.
DISCUSSION. The term “nuclear pharmacy” is applied to a special unit of a medical organisation where RFLP synthesis and quality control is carried out. The practice of foreign hospital nuclear pharmacies has proven their advantages in terms of ensuring the shortest route of vital drugs to patients. The article analyses the main requirements of Russian regulatory documents and recommendations on the functioning of a nuclear pharmacy, as well as the experience of the first nuclear pharmacy based on A.F. Tsyb Medical Radiological Research Center — Branch of the National Medical Research Center of Radiology of the Ministry of Health of the Russian Federation. Regular work of the radiopharmacy allows to treat up to several hundred patients with vital but not yet registered drugs. However, nuclear pharmacies are far from being a routine practice for Russia, and
a number of issues has to be addressed to allow for a proper function of these pharmacies: creating a training system for radiopharmacists; defining the scope of preliminary studies of RPs extemporaneously produced at a pharmacy; updating radiation safety documents regulating process launch for new radionuclides at a pharmacy, and so on.
CONCLUSIONS. The world practice of hospital radiopharmacies has proven this system effective for RP commerce. Globally, approaches to RP preparation may be defined at the national or regional level. Pharmacies authorised to prepare RPs is a new type of pharmacy not previously known in the EAEU Member states. The creation of a regulatory framework for RP preparation should accommodate world practices with proven efficiency. The high-priority tasks to be addressed are training specialised personnel; revising a variety of documents on radiation safety; creating an expert panel assisting numerous issues associated with developing radiopharmacies; and obtaining funding sources for RP treatment.
INTRODUCTION. Current risk-based general strategies for the control of elemental impurities in medicinal products, as documented in the elemental impurity monographs of national and world pharmacopoeias, do not apply to radiopharmaceuticals. Manufacturers of radiopharmaceuticals have to determine and substantiate the lists of specified elemental impurities and their limits.
AIM. This study aimed to develop a control strategy for elemental impurities in radiopharmaceuticals by analysing the requirements of national and world pharmacopoeias to the levels of specified elemental impurities in radiopharmaceuticals.
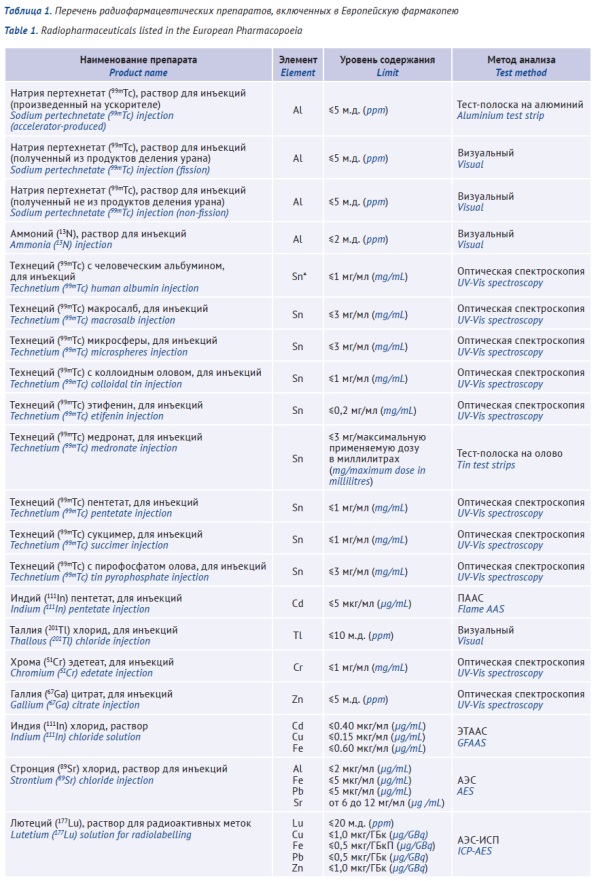
DISCUSSION. Limits for the amounts of elemental impurities in specific radiopharmaceuticals are provided in individual monographs of pharmacopoeias (24 monographs in the European Pharmacopoeia, 7 monographs in the Indian Pharmacopoeia, 7 monographs in the State Pharmacopoeia of the Russian Federation, 6 monographs in the United States Pharmacopeia, 6 monographs in the Japanese Pharmacopoeia, 6 monographs in the Pharmacopoeia of the People’s Republic of China, and 4 monographs in the Korean Pharmacopoeia). Individual monographs for the same radiopharmaceuticals differ in the lists of elemental impurities and the corresponding limits. Moreover, these monographs lack common criteria for selecting elemental impurities and establishing limits for them. In most cases, limits are set for the parent non-radioactive elements, the elements that are added as components of radiolytic stabilisers or sorbents (Ti, Zr, Sn, and Al oxides), and the elements that decrease radiolabelling efficiency (Cu, Fe, Pb, and Zn in 177Lu solutions; Cu, Cd, and Fe in 111InCl solutions; Cd, Cu, Fe, Pb, and Zn in 90Y solutions; etc.). Elemental impurity limits for radiopharmaceuticals are calculated on the basis of the acceptable impact on the physicochemical, biological, and radiochemical properties of the product used and the dose administered. The elements that lack specified permitted daily exposure (PDE) limits are subject to specification if these elements affect the target organ or the process of medicinal product radiolabelling. There is also no unified approach to limiting the amounts of elemental impurities in radiopharmaceutical precursors.
CONCLUSIONS. This article provides radiopharmaceutical manufacturers with a control strategy for elemental impurities in finished medicinal products, based on assessing the risk of elemental impurities having a negative impact on the quality attributes of medicinal products. This strategy includes generalised criteria for selecting the list of specified impurities and their limits for radiopharmaceuticals, which significantly streamlines the preparation of regulatory documentation concerning this quality attribute.
INTRODUCTION. Radiopharmaceutical manufacturers should assess potential negative impact of elemental impurities on the quality of their products using real data of their elemental contamination. Accumulating knowledge about medicinal products and their manufacturing process should start from the development stage and continue throughout the implementation stage until production finally ends.
AIM. This study aimed to monitor elemental impurities content in Russian radiopharmaceuticals using inductively coupled plasma mass spectrometry.
MATERIALS AND METHODS. The samples included 127 Russian radiopharmaceuticals obtained by the Scientific Center for Expert Evaluation of Medicinal Products during its statutory activities: fluorodeoxyglucose (18F), PSMA-1007 (18F), sodium iodide (131I), sodium iodohippurate (131I), iobenguane (123I), and sodium pertechnetate (99mTc). The content of Ag, Al, As, Au, Ba, Be, Bi, Cd, Co, Cr, Cu, Fe, Ga, Ge, Hg, Ir, Li, Mn, Nb, Ni, Os, Pb, Pd, Pt, Rh, Ru, Sb, Se, Mo, Sn, Tl, V, W, and Zn was detected on the Agilent 7900 mass spectrometer.
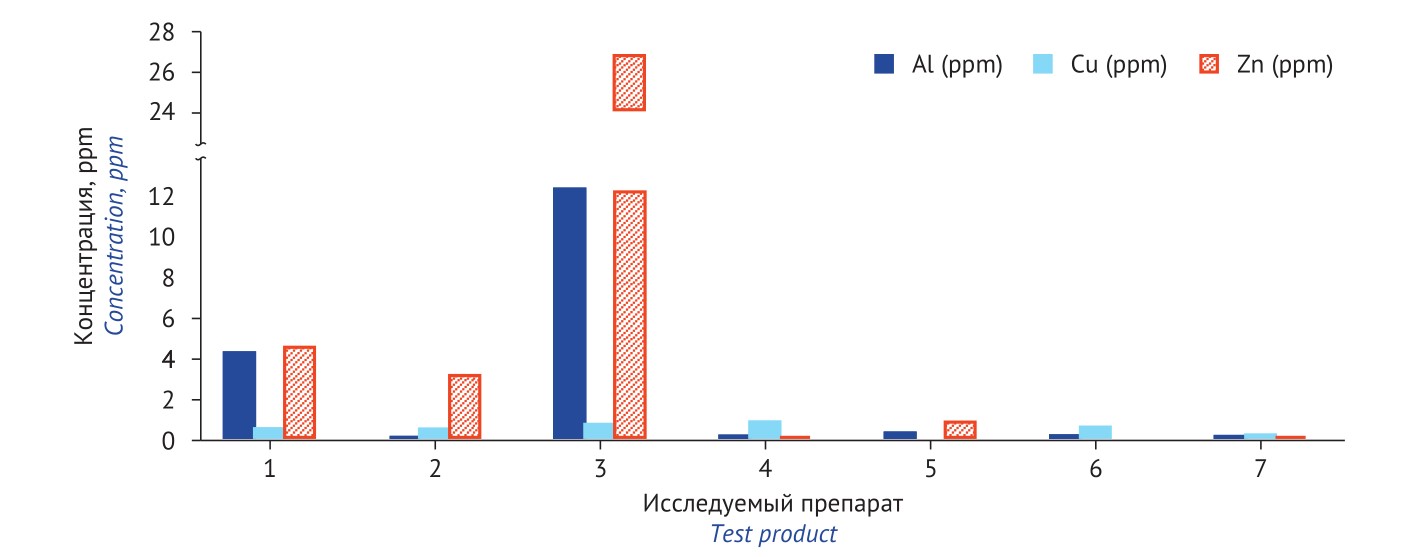
RESULTS. Elemental impurities Ag, As, Au, Bi, Be, Cd, Co, Cr, Ga, Ge, Hg, Ir, Mo, Nb, Ni, Os, Pb, Pd, Pt, Rh, Ru, Sb, Se, Sn, Tl, V, and W were not detected in any of the tested radiopharmaceuticals. Al, Zn, and Cu mostly contributed to elemental contamination in almost all radiopharmaceuticals. Radiopharmaceuticals used for single-photon emission computed tomography (SPECT) contained significantly less elemental impurities compared to positron emission tomography (PET) radiopharmaceuticals.
CONCLUSIONS. The elements that mostly contaminate Russian radiopharmaceuticals are non-toxic or low-toxic (Al, Zn, Cu). These impurities should be monitored in case they negatively impact the quality of radiopharmaceuticals. In fluorodeoxyglucose (18F), elemental contamination heavily depends on the used buffer solution. In order to find out elemental contamination sources in the studied radiopharmaceuticals, an additional analysis of impurities in the raw materials, reagents and semi-products is indispensable at all technological stages.
INTRODUCTION. When developing and validating manufacturing of a product, Russian radiopharmaceutical manufacturers are required to detect ubiquitous impurities (Pb, As, Fe as well as other elements) causing contamination. At the stage of preliminary monitoring, Al, Cu, and Zn were found to most often contribute to fluorodeoxyglucose (18F) elemental contamination.
AIM. This study aimed to develop and validate a technique detecting Al, As, Cu, Fe, Pb, and Zn in fluorodeoxyglucose (18F) using inductively coupled plasma mass spectrometry (ICP-MS).
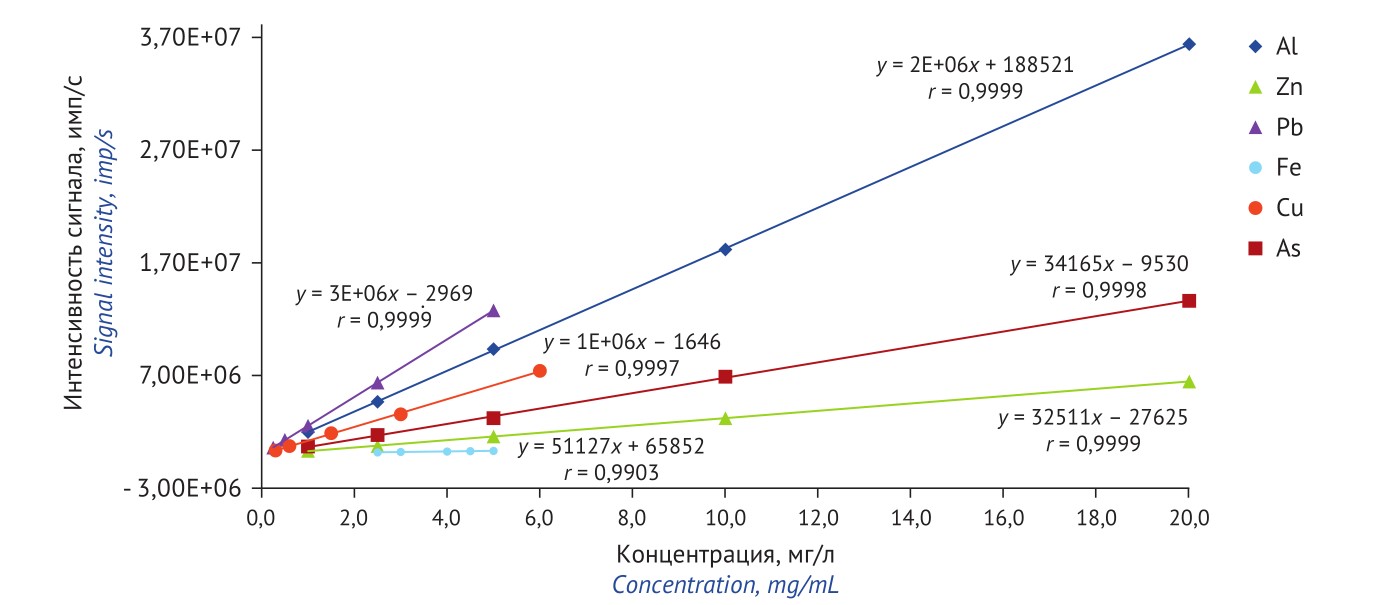
MATERIALS AND METHODS. The technique was developed on a model mixture simulating fluorodeoxyglucose (18F). A sample with minimum analysed impurities was used for validation; then varying amounts of standard analyte solutions were added. The content was determined using Agilent 7900 mass spectrometer. Signal intensities of the following isotopes (a.u.m.) were recorded: 27Al, 57Fe, 63Cu, 66Zn, 75As, and 208Pb.
RESULTS. The organic matrix was found to insignificantly affect elemental analysis. Due to matrix effect, NaCl reduced spike recovery of Al, Cu, Pb, and Zn to the level below 70%. When diluting model mixture tenfold, spike recovery of the analytes reached 90–101%. Correlation coefficients of element concentration — signal intensity linear dependences for the analytes exceeded 0.99. When estimating precision, RSD values did not exceed 10%.
CONCLUSIONS. A technique has been developed detecting Al, As, Cu, Fe, Pb, and Zn in fluorodeoxyglucose (18F) using ICP-MS. This technique considers influence of matrix components on quantification of target elements when preparing samples. Validation parameters of the technique comply with pharmacopoeial requirements.
EXPERT OPINION
INTRODUCTION. Radiopharmaceuticals (radiopharmaceutical medicinal products) are used in clinical practice to diagnose and treat a wide range of diseases. However, regulatory acts of the Eurasian Economic Union give no detailed requirements for a section dedicated to clinical documentation in the authorisation dossier currently developed as Common Technical Document (CTD) used to assess safety and effectiveness of this drug category.
AIM. This study aimed to identify transparent principles used to draft clinical modules of authorisation dossiers for various types of radiopharmaceuticals based on analysed provisions of the current EAEU legislation, recommendations by international regulatory authorities, and the expertise.
DISCUSSION. Drug authorisation in the EAEU and adjusting national authorisation dossiers with the broader EAEU requirements (including new countries of recognition) requires a full dataset on clinical studies of radiopharmaceuticals. Radiopharmaceuticals have a whole range of specific traits, both due to the nature of these products and their clinical use. Specific traits of radiopharmaceuticals shall be thoroughly studies and described in the dossier documents. This study analysed the current EAEU regulatory documents and recommendations given by: Swiss Agency for Therapeutic Products (Swissmedic) and Canada Ministry of Health (Health Canada). The authors developed possible scenarios of submitting full dossiers required for a radiopharmaceutical, according to their type / kind and clinical use.
CONCLUSIONS. The methods described to develop dossiers for various types / kinds of radiopharmaceuticals will help drug developers and regulatory affairs managers fully comply with the authorisation procedures.
PHARMACOPOEIA
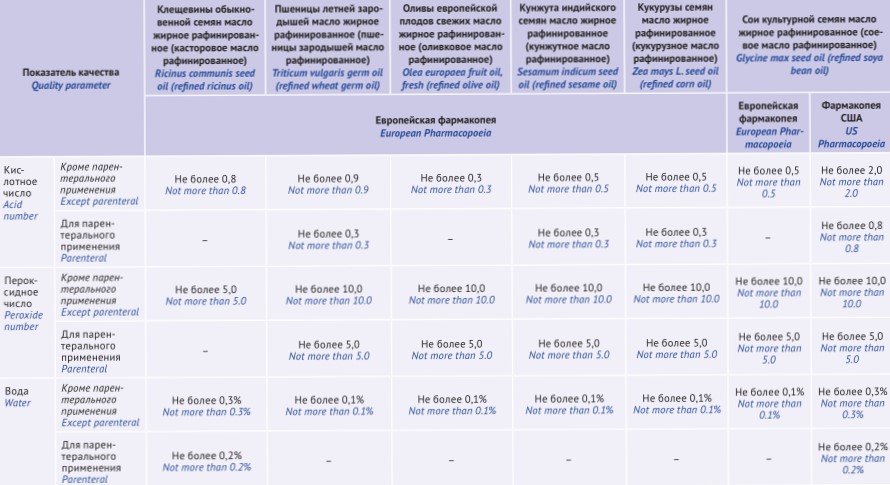
INTRODUCTION. Vegetable fatty oils are widely used in modern pharmaceutical practice. Depending on production and purification method, they can serve as an active pharmaceutical substance and/or as an excipient. Quality and nomenclature requirements for vegetable fatty oils result from their production and purification methods.
AIM. This study aimed to harmonise national quality standards for fatty vegetable oils with the requirements of the world leading pharmacopoeias.
DISCUSSION. Aligning general pharmacopoeial monographs (GPM) with the standards of the leading world pharmacopoeias, as well as regional EAEU Pharmacopoeia is important for standardisation of biologically active compounds in order to align requirements of various pharmacopoeias. According to a comparative analysis of the leading world pharmacopoeias, the European (Eur. Pharm.) and British (BP) pharmacopoeias describe classification, production methods and purification of fatty vegetable oils. Purification allows obtaining a certain quality of oils used, among others, for production of parenteral drugs. Unlike Eur. Pharm. and BP monographs describing only production and purification methods, Japanese (JP) and United States (USP) pharmacopoeias provide identification and test methods applicable to fatty vegetable oils.
CONCLUSIONS. The analysed monographs from the leading world pharmacopoeias were used to develop a GPM draft “Vegetable Fatty Oils” harmonised with similar requirements of the leading world pharmacopoeias. The common approach classifies oils depending on production and purification method, which affects their quality and further medical use. More rigorous requirements are considered for oils intended for parenteral drugs and listed in the relevant monographs.
DEVELOPMENT OF MEDICINES
INTRODUCTION. Major international pharmaceutical companies play a crucial role in the development of original medicines. To determine the directions for original medicines development in Russia, it is essential to analyse global trends and emerging weak signals (tendencies) — early indicators of future-significant innovations. OBJECTIVE. To identify trends and emerging weak signals that could shape pharmaceutical development in Russia through an analysis of the pipeline of original medicines being developed by global pharma leaders.
MATERIALS AND METHODS. An analysis was conducted on the medicinal product development plans for 2024 of the 20 largest pharmaceutical companies globally, which have the highest research and development budgets. The focus was on original medicines scheduled for clinical trials Phase 1 from January to May 2024. A descriptive research approach was applied, based on retrospective analysis of secondary data. The study measured the number of original medicines under development, research directions, target classes, medicine types, and groups. Both quantitative and qualitative evaluations were used to identify key trends and tendencies (emerging weak signals) in pharmaceutical development of medicines.
RESULTS. During the analysed period, 17 out of 20 leading pharmaceutical companies initiated Phase 1 trials for a total of 84 original medicines. The most active research areas included oncology, endocrinology and metabolism, cardiovascular system, and immunology. Notably, 40 medicines entered Phase 1 trials in oncology. The largest share (42%) of the medicines in development consists of high molecular weight molecules. Based on the number of medicines developed by multiple companies, trends were identified for the following medicine classes: “Large molecule” — bispecific antibodies (10 medicines, 5 developers); monospecific antibodies (8 medicines, 7 developers); antibody-drug conjugates (8 medicines, 3 developers); “Small molecule” — enzyme inhibitors (9 medicines, 6 developers); “Cell therapy” — CAR-T-based therapies (6 medicinal products, 2 developers).
CONCLUSIONS. Current trends in targeted therapy development include the creation of bispecific antibodies and next-generation antibody-drug conjugates, alongside CAR-T therapies based on autologous T cells, predominantly for the treatment of malignant neoplasms. The study of multispecific antibodies is shaping a new direction in targeted cancer therapy. The development of low-molecular-weight enzyme inhibitors is establishing a trend in various therapeutic areas. Specifically, enzyme inhibitors targeting synthetic lethal vulnerabilities like WRN and PRMT5 are emerging as a key tendency in small-molecule medicine development for targeted cancer therapy.
ISSN 3034-3453 (Online)